Cycling Webquest
1. Draw the carbon cycle (on a separate piece of paper)
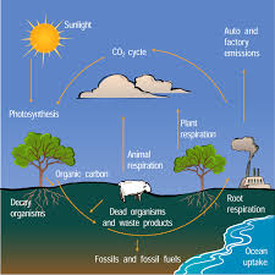
2. How does carbon exist in the atmosphere?
Carbon is an element that you an find in any living organism.
3. How are fossil fuels created? Explain.
Some that are buried underground and will become fossil fuels in millions of years. Then these fossil fuels are burned and made into gasoline
4. Describe two ways that carbon enters the atmosphere.
Carbon is attached to oxygen in a gas called carbon dioxide. With the help of the Sun, through the process of photosynthesis, carbon dioxide is pulled from the air to make plant food from carbon
5. How are the oceans involved in the carbon cycle?
The oceans, and other bodies of water, soak up some carbon from the atmosphere.
6. How is the temperature of the Earth partly controlled by carbon?
Carbon dioxide is a greenhouse gas and traps heat in the atmosphere. Without it and other greenhouse gases, Earth would be a frozen world.
7. What role do rocks have within the carbon cycle?
Rocks on land can add carbon to surface water which eventually runs off to the ocean.
8. Where are you starting within the carbon cycle?
A carbon atom
9. How much of the atmosphere is made of carbon dioxide (CO2)?
(0.04%)
10. By how much has CO2 increased in the atmosphere during the past 150 years?
Due to burning fossil fuels, the amount has increased 30% in the past 150 years.
11. Next stop = Land Plants
What did you learn?
As more carbon dioxide is added to our atmosphere, plants will
be able to grow faster.
12. Next stop = Soil
What did you learn?
Soils store about 3% of Earth's carbon. As bacteria and fungi breakdown the detritus, carbon is sent into the atmosphere.
How much carbon does the surface ocean absorb from the atmosphere each year?
A:The surface ocean takes in approximately 90 Gigatons of carbon per year.
True or False: When plants die and decay, they bring carbon into soil.
True
13. Next stop = Surface Oceans
What did you learn?
The ocean absorbs more carbon dioxide from the atmosphere than the land does.
14. Next stop = Deep Ocean Currents
What did you learn?
The deep ocean gets carbon from circulation with the surface ocean and dead and decaying marine life.
15. Next stop = Marine Animals
What did you learn?
Marine life cannot survive without carbon.
Q:When carbon enters the deep ocean, how long does it stay there?
Hundreds of years
True or False: Phytoplankton are tiny plants and algae that float in the ocean and take up carbon dioxide as they grow.
True
True or False: Plants both absorb CO2 from the atmosphere and release it into the atmosphere.
True
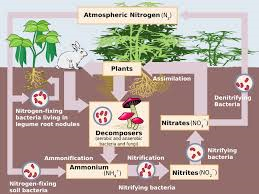
16.What are the two conditions under which nitrogen will react with oxygen? (In other words, what is necessary for nitrogen in the air to combine with oxygen?)
A: The two conditions : high temperature and pressure lightning bolts give
17. What are the two compounds that are formed when nitrogen combines with oxygen?
Nitric oxide:
Nitrogen dioxide
18. How does nitric acid (HNO3) form?
Nitrogen dioxide react with rain water.
19. Why is nitric acid (HNO3) important?
Nitric acid provides plant's nutrients.
20. What percentage of the air we breathe is nitrogen?
79 percent of the air is nitrogen.
21. Even though considerable
nitrogen is available in the air, most plants do not use the
nitrogen
(N2) found in the air. Why not?
Plants find it not really usable because nitrogen then turns into ammonia which is already released from plants.
22. In what compounds can plants use nitrogen? They can only use nitrogen when this is in a fixed form, such as:
Nitrate ions and Ammonium ions
23. How do animals get the nitrogen they need?
Animals eat plants that basically use the nitrogen system to grow.
24. Atmospheric nitrogen (N2) is stably put together. It does not break apart easily. If it can't be broken easily, organisms definitely will not find it easy as well. Nitrogen fixation is used to break up nitrogen.
a. What is atmospheric fixation?
The enormous energy of lightning breaks nitrogen molecules and enables their atoms to combine with oxygen in the air forming nitrogen
b. What is industrial fixation? [This is how artificial fertilizers are made.]
Industrial fixation is a process in where it is done unnaturally by us humans and it is to break down nitrogen molecules.
c. What is biological fixation? (In your answer, describe the types of plants associated with the symbiotic relationship.)
It is only found in some bacteria.
25. Draw the nitrogen cycle: On a separate piece of paper: (Remember there are other diagrams on the previous websites.) If you’re not sure what a term means, look through the reading and links for help.
(Picture is Below)
)0))))tytrvertrtg BBBdjbdhrejsgof suriBelow )b
2. How does carbon exist in the atmosphere?
Carbon is an element that you an find in any living organism.
3. How are fossil fuels created? Explain.
Some that are buried underground and will become fossil fuels in millions of years. Then these fossil fuels are burned and made into gasoline
4. Describe two ways that carbon enters the atmosphere.
Carbon is attached to oxygen in a gas called carbon dioxide. With the help of the Sun, through the process of photosynthesis, carbon dioxide is pulled from the air to make plant food from carbon
5. How are the oceans involved in the carbon cycle?
The oceans, and other bodies of water, soak up some carbon from the atmosphere.
6. How is the temperature of the Earth partly controlled by carbon?
Carbon dioxide is a greenhouse gas and traps heat in the atmosphere. Without it and other greenhouse gases, Earth would be a frozen world.
7. What role do rocks have within the carbon cycle?
Rocks on land can add carbon to surface water which eventually runs off to the ocean.
8. Where are you starting within the carbon cycle?
A carbon atom
9. How much of the atmosphere is made of carbon dioxide (CO2)?
(0.04%)
10. By how much has CO2 increased in the atmosphere during the past 150 years?
Due to burning fossil fuels, the amount has increased 30% in the past 150 years.
11. Next stop = Land Plants
What did you learn?
As more carbon dioxide is added to our atmosphere, plants will
be able to grow faster.
12. Next stop = Soil
What did you learn?
Soils store about 3% of Earth's carbon. As bacteria and fungi breakdown the detritus, carbon is sent into the atmosphere.
How much carbon does the surface ocean absorb from the atmosphere each year?
A:The surface ocean takes in approximately 90 Gigatons of carbon per year.
True or False: When plants die and decay, they bring carbon into soil.
True
13. Next stop = Surface Oceans
What did you learn?
The ocean absorbs more carbon dioxide from the atmosphere than the land does.
14. Next stop = Deep Ocean Currents
What did you learn?
The deep ocean gets carbon from circulation with the surface ocean and dead and decaying marine life.
15. Next stop = Marine Animals
What did you learn?
Marine life cannot survive without carbon.
Q:When carbon enters the deep ocean, how long does it stay there?
Hundreds of years
True or False: Phytoplankton are tiny plants and algae that float in the ocean and take up carbon dioxide as they grow.
True
True or False: Plants both absorb CO2 from the atmosphere and release it into the atmosphere.
True
16.What are the two conditions under which nitrogen will react with oxygen? (In other words, what is necessary for nitrogen in the air to combine with oxygen?)
A: The two conditions : high temperature and pressure lightning bolts give
17. What are the two compounds that are formed when nitrogen combines with oxygen?
Nitric oxide:
Nitrogen dioxide
18. How does nitric acid (HNO3) form?
Nitrogen dioxide react with rain water.
19. Why is nitric acid (HNO3) important?
Nitric acid provides plant's nutrients.
20. What percentage of the air we breathe is nitrogen?
79 percent of the air is nitrogen.
21. Even though considerable
nitrogen is available in the air, most plants do not use the
nitrogen
(N2) found in the air. Why not?
Plants find it not really usable because nitrogen then turns into ammonia which is already released from plants.
22. In what compounds can plants use nitrogen? They can only use nitrogen when this is in a fixed form, such as:
Nitrate ions and Ammonium ions
23. How do animals get the nitrogen they need?
Animals eat plants that basically use the nitrogen system to grow.
24. Atmospheric nitrogen (N2) is stably put together. It does not break apart easily. If it can't be broken easily, organisms definitely will not find it easy as well. Nitrogen fixation is used to break up nitrogen.
a. What is atmospheric fixation?
The enormous energy of lightning breaks nitrogen molecules and enables their atoms to combine with oxygen in the air forming nitrogen
b. What is industrial fixation? [This is how artificial fertilizers are made.]
Industrial fixation is a process in where it is done unnaturally by us humans and it is to break down nitrogen molecules.
c. What is biological fixation? (In your answer, describe the types of plants associated with the symbiotic relationship.)
It is only found in some bacteria.
25. Draw the nitrogen cycle: On a separate piece of paper: (Remember there are other diagrams on the previous websites.) If you’re not sure what a term means, look through the reading and links for help.
(Picture is Below)
)0))))tytrvertrtg BBBdjbdhrejsgof suriBelow )b
26. Why is nitrogen needed by plants and animals?
This is because it is the foundation of living. Plants don't need as much consumption of nitrogen as animals do because plants have nitrogen in them already.
Animals then eat these plants and that's how they are able to survive. It helps with the strength, power, growth, and metabolism they have.
This is because it is the foundation of living. Plants don't need as much consumption of nitrogen as animals do because plants have nitrogen in them already.
Animals then eat these plants and that's how they are able to survive. It helps with the strength, power, growth, and metabolism they have.