Properties of Water
- An atom is the smallest unit matter that retains its properties.
- Atoms consist of Protons, Electrons, and Neutrons.
- Protons have a positive charge, while Electrons have a negative, and Neutrons have no charge.
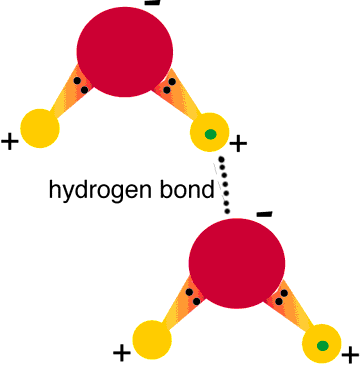
- Water is a polar molecule.
- Hydrogen Bonding is the weak attraction between the weekly negative oxygen atoms of a different molecule.
- Water is a universal solvent.
- Cohesion is when water take the shape of different objects.
- Surface Tension is when water is attracted to itself.
- Adhesion is water molecules attracted to other materials.
- Heat is a measure of total kinetic energy of the atoms and molecules
Investigating Water Lab

Evaporate Cooling: station #1 Water and Alcohol
When I tested both the Alcohol and water the alcohol
felt much cooler then the water on my hand.
When I tested both the Alcohol and water the alcohol
felt much cooler then the water on my hand.

Water Tension/Cohesion- station #2 Water on a Penny
I predicted
about 45 drops of water would fit on the penny, but only 30
drops were able
to fit until the surface tension broke.
I predicted
about 45 drops of water would fit on the penny, but only 30
drops were able
to fit until the surface tension broke.

Surface Tension: Station #3 Water on Wax Paper
During this station I observed that when I dropped water or alcohol
on the wax paper the water did not bleed through like it would with
regular paper. I think this is probably because of surface tension.
During this station I observed that when I dropped water or alcohol
on the wax paper the water did not bleed through like it would with
regular paper. I think this is probably because of surface tension.

Surface Tension Station #4- Paper Clips on the Surface of a cup water
During this station I was confident that I could hold 5 paper
clips at the surface water but it turned out I could only make 1 float. I wasn't
so careful when placing the paperclip so all of them kept
sinking.
During this station I was confident that I could hold 5 paper
clips at the surface water but it turned out I could only make 1 float. I wasn't
so careful when placing the paperclip so all of them kept
sinking.

Adhesion: Station #5- Graduated Cylinder
The reason why the water line in the graduated cylinder wasn't straight was because due
to adhesion the water was sticking to the glass.
The reason why the water line in the graduated cylinder wasn't straight was because due
to adhesion the water was sticking to the glass.

Capillary Action- Station #6- Rolled up Paper Towel
Capillary action is important because it gives the
plants and trees the water they need to survive. The food coloring transferred
to the other side of the cup just like water does when its transferred from the
roots to a plant.
Capillary action is important because it gives the
plants and trees the water they need to survive. The food coloring transferred
to the other side of the cup just like water does when its transferred from the
roots to a plant.
Introduction to Density Lab
Materials:
For our first step of the procedure I weighed the Beaker 1 and Beaker 2 by themselves. Beaker 1 weighed 111.7g and Beaker 2 weighed 110.1g. I then measured 250 ml of cold water in beaker 1 and 250 ml in beaker 2. Next I added 20g of salt and 5 drops of blue food coloring. I next added 5 drops of yellow food coloring to the warm fresh water. I recorded the new masses of the beakers and ended up with 212.7g for beaker 1 and 191.3g for beaker 2.
- Two 250 ml beakers, one 500ml beaker
- Electronic scale
Glass stirring rod, spon - Pickling Salt, Water samples (warm and cold), and food coloring (yellow and blue)
- ipad
For our first step of the procedure I weighed the Beaker 1 and Beaker 2 by themselves. Beaker 1 weighed 111.7g and Beaker 2 weighed 110.1g. I then measured 250 ml of cold water in beaker 1 and 250 ml in beaker 2. Next I added 20g of salt and 5 drops of blue food coloring. I next added 5 drops of yellow food coloring to the warm fresh water. I recorded the new masses of the beakers and ended up with 212.7g for beaker 1 and 191.3g for beaker 2.
For part 2 I Poured warm, fresh water into the 500 ml beaker. I then poured the cold salt water into the beaker. During this experiment I observed that the layers were starting to settle down from the beaker.