Save Our Shells/ Ocean Acidification
Bubbles Protocol
Pre-Lab Questions:
1. What gas are you blowing into the water?
Carbon Dioxide.
2. What happens to the gas when you blow it into water?
Dissolves or absorbs into the water.
3. How are you measuring the pH of the water during this lab?
The pH (color)
4. What does measuring the pH of the water tell us?
How much carbon dioxide is being absorbed.
5. After studying the reactions above, how do you think carbonic acid will affect the pH of salt water?
It will be more acidic
Hypothesis: If I blow or add CO2 to seawater, then it will change the pH.
Protocol:
Control Trial: The control trial does not change during the experiment. We added 100ml of saltwater to a 500ml beaker. After to find out if the color changes or not, we record the initial color using the pH table. We placed a white piece of paper underneath. We put some saran on top of the beaker to cover it. We inserted the straw by poking the hole through the saran wrap. My partner Omar (Omaru) blows in the straw. We set the timer for 2 minutes and record the results every 30 seconds.
Pre-Lab Questions:
1. What gas are you blowing into the water?
Carbon Dioxide.
2. What happens to the gas when you blow it into water?
Dissolves or absorbs into the water.
3. How are you measuring the pH of the water during this lab?
The pH (color)
4. What does measuring the pH of the water tell us?
How much carbon dioxide is being absorbed.
5. After studying the reactions above, how do you think carbonic acid will affect the pH of salt water?
It will be more acidic
Hypothesis: If I blow or add CO2 to seawater, then it will change the pH.
Protocol:
Control Trial: The control trial does not change during the experiment. We added 100ml of saltwater to a 500ml beaker. After to find out if the color changes or not, we record the initial color using the pH table. We placed a white piece of paper underneath. We put some saran on top of the beaker to cover it. We inserted the straw by poking the hole through the saran wrap. My partner Omar (Omaru) blows in the straw. We set the timer for 2 minutes and record the results every 30 seconds.
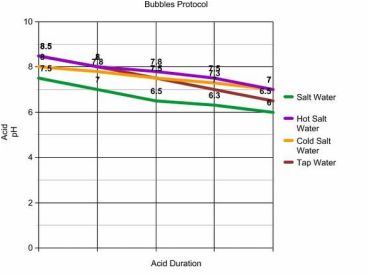
Yes my conclusion was right. The pH changed colors over
a certain amount of time. I learned that over time, acid still increases if
carbon dioxide is added into that substance.
a certain amount of time. I learned that over time, acid still increases if
carbon dioxide is added into that substance.
Save Our Shells
Central Question: How does a decrease in the pH of seawater affect the calcium carbonate shells of animals?
Student Pre-Lab
Questions:
1. How do organisms make their shells? What are shells made of?
CaCo3
2. What do you expect to happen to the shell in an acidic solution such as vinegar?
the shell will rust and it will cause the pH levels in the water to change due to the different floating materials.
3. What are the sources of carbon dioxide and which of these sources are most likely to affect ocean pH?
Fossil fuels
Hypothesis: If we put mussel shells in the acidic water, they will become more venerable.
(Pictures Below)
Central Question: How does a decrease in the pH of seawater affect the calcium carbonate shells of animals?
Student Pre-Lab
Questions:
1. How do organisms make their shells? What are shells made of?
CaCo3
2. What do you expect to happen to the shell in an acidic solution such as vinegar?
the shell will rust and it will cause the pH levels in the water to change due to the different floating materials.
3. What are the sources of carbon dioxide and which of these sources are most likely to affect ocean pH?
Fossil fuels
Hypothesis: If we put mussel shells in the acidic water, they will become more venerable.
(Pictures Below)